SOFTER
SOFTER aims to develop a next-generation Tumor-on-Chip (ToC) model that recapitulates the tumor microenvironment (TME) of endometrial cancer (EC), a gynecological malignancy projected to become the third most common cancer and fourth leading cause of cancer deaths among women by 2030. EC originates from endometrial hyperplasia and progresses within the complex architecture of the endometrium, which includes epithelial, stromal, endothelial, and immune cells. Despite the availability of surgery and adjuvant therapies for many patients, treatment options remain limited and often ineffective in advanced or recurrent EC. Notably, immunotherapy has shown promise, particularly for patients with microsatellite instability (MSI), yet the majority (75%) of EC cases are microsatellite stable (MSS) and exhibit poor response to such treatments. Therefore, there is an urgent need for advanced preclinical models that can predict individual treatment responses and facilitate the development of innovative therapies for the MSS population.
Coordinator

Professor
Université Claude Bernard Lyon 1
Institutions and establishments involved
Work Packages
CNRS; Inserm; Université Claude Bernard Lyon 1; Centre Léon Bérard (CLB), Hospice Civiles de Lyon (HCL)
To address this challenge, SOFTER proposes the creation of a Soft-ToC platform specifically designed to mimic key features of the EC TME: hypoxia, a tunable extracellular matrix (ECM) stiffness, and the presence of vascular and immune components. This integrated platform will be developed through four interconnected work packages (WPs):
- WP1: Collection, characterization, and biobanking of patient-derived EC tissues as tumoroids, with a focus on the MSS subtype.
- WP2: Engineering of the Soft-ToC device, incorporating ECM-mimicking hydrogels, oxygen gradients, endothelialized channels to simulate vascularization, and resident immune cell integration—key components that influence response to immunotherapies.
- WP3: Development of a robust spatio-temporal imaging and analysis pipeline to monitor EC cell behavior in real time, extracting key biological (growth, invasion, migration) and biophysical (micro-elastography, Brillouin microscopy) biomarkers over 8 days. This will be complemented by in toto immunostaining and single-cell RNA sequencing to track the evolution of cellular subpopulations within the Soft-ToC.
- WP4: Evaluation of therapeutic response using standard chemotherapies (Paclitaxel, Carboplatin) and immunotherapies (POC done on TLR ligands as adjuvants). For standard chemotherapies, the Soft-ToC’s predictive performance will be benchmarked against
tumoroids and validated through correlation with patient outcome data. Deep Learning approaches will be used to correlate multi-parametric readouts with clinical outcomes and identify robust biomarker signatures.
Research program
Technologically, SOFTER represents a significant step forward in the evolution of microfluidic cancer models. By combining multiplexing, automation readiness, and physiological relevance, the platform aims to meet the reproducibility and scalability standards required for translational research and preclinical drug testing. It addresses major bottlenecks in the adoption of Organ-on-Chip technologies, especially in oncology, where patient-derived systems incorporating mechanical, immunological, and hypoxic cues are still rare.
From a translational perspective, SOFTER offers a powerful tool for personalized medicine. It enables predictive assessments of patient-specific treatment responses, including those to novel immunotherapeutics, in a controlled and reproducible in vitro environment. Furthermore, by integrating deep learning to analyze complex datasets generated from the platform, SOFTER will provide actionable insights into treatment efficacy and resistance mechanisms, supporting its adoption by pharmaceutical and clinical research communities. Aligned with the 4R principles (Replace, Reduce, Refine, Reuse animal models), SOFTER offers an ethical, cost-effective, and predictive alternative to animal testing, accelerating the development of precision oncology solutions for endometrial cancer.


Expected outcomes
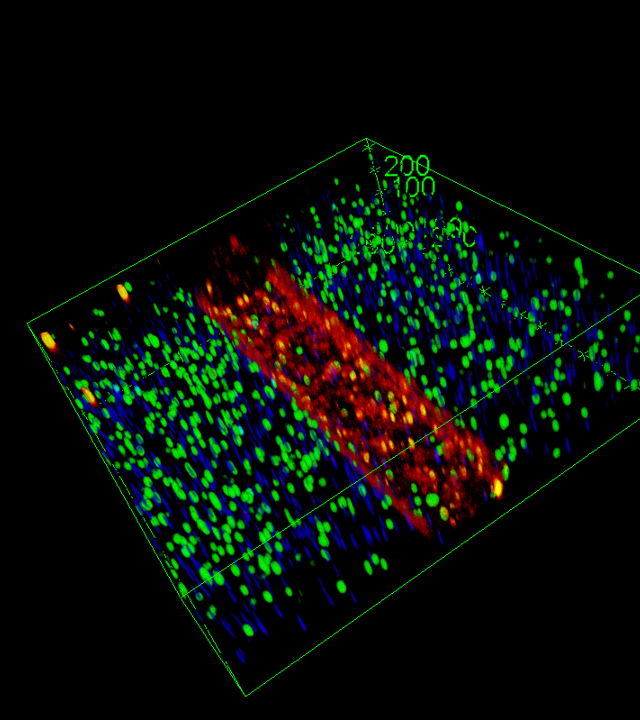
SOFTER aims to develop an advanced Soft-ToC with better predictive potential than conventional assays. SOFTER will be specifically designed to mimic the major features of EC tumor microenvironment: hypoxia, a dense ECM matching EC rigidity, as well as resident
immune cells and endothelialized channels (Fig.1). By performing a spatiotemporal analysis of patient-derived cell response, the relevance and predictive potential of the SOFTER platform will be proof-tested with classical chemotherapeutic treatments, as well as IT with immune adjuvants. Ultimately, Deep Learning approaches will be used to correlate SOFTER-derived data with patient outcomes, allowing the identification of a subset of relevant biological and biophysical biomarkers, and paving the way for its use in personalized medicine and therapeutic decision-making.
The consortium
SOFTER will build on the consortium already established through a funded project (Ectochip, ShapeMed@Lyon), bringing together an interdisciplinary team to develop an innovative Tumor-on-Chip (ToC) model for endometrial cancer (EC), capable of reproducing the cellular and mechanical properties of the tumor as well as its immune microenvironment.
Patient recruitment for the project is made possible through agreements already established by the 3D-ONCO platform at CRCL with the HCL and the CLB, and relies on the clinical expertise in endometrial cancer of Prof. I. Ray-Coquard and Dr. C.A. Philipp, as well as on the expertise of the 3D-ONCO platform in generating patient-derived organoid models.
SOFTER will also rely on complementary key expertise, notably in immunology and the immune microenvironment (J. Valladeau-Guillemond, CRCL, Department of Immunology), endothelialization (C. Dessalles), hypoxic environments (J.-P. Rieu), biophysical measurements (T. Dehoux, S. Catheline), and machine learning (O. Cochet-Escartin).
The feasibility of the project is supported by key preliminary results obtained by the partner teams, which also contribute to reducing associated risks.
Institut Lumière Matière; Centre Léon Bérard; Centre de Recherche en Cancérologie de Lyon; Laboratoire des Applications Thérapeutiques des Ultrasons.
Plus de projets