ON-CHIP
Glaucoma, particularly primary open-angle glaucoma (POAG), is a multifactorial neurodegenerative disease resulting from complex interactions between intraocular pressure, neuroinflammation and genetic factors. It causes progressive and irreversible degeneration of retinal ganglion cells (RGCs), whose axons form the optic nerve, for which therapeutic options remain limited. The lack of an OAG model that integrates the various components of the disease limits the development of innovative therapies. Among these, cell replacement therapies offer real hope for patients. On-chip will overcome therapeutic obstacles such as the limited survival and integration of transplanted cells in an altered nervous environment.
Coordinator

Assistant Professor
Inserm
Institutions and establishments involved
Work Packages
CNRS; Inserm; Sorbonne Université / IBPS (Neuro-SU)
The project is structured around three interconnected objectives:
- WP1: Design and fabricate the optic nerve-on-a-chip device, incorporating unidirectional
connected retinal to brain organoids and key cellular components involved in ONH
homeostasis. - WP2: Model glaucoma by exposing the system to POAG-relevant stressors, analyzing their
effects on RGC function and survival. - WP3: Assess therapeutic strategies, including iPSC-derived cell transplantation, gene
therapy, and neuroimmunomodulation to improve graft integration
Research program
This project proposes the development of an innovative Human Optic Nerve-on-a-Chip platform, a multi-organ microfluidic device designed to address the urgent need for physiologically relevant models in glaucoma research. Glaucoma is one of the leading causes of blindness in adults and is characterised by the progressive degeneration of retinal ganglion cells (RGCs), which collect the visual information captured and processed by the retina and transmit it to the brain via their axons forming the optic nerve. This cellular loss results from a combination of mechanical, inflammatory and genetic factors.
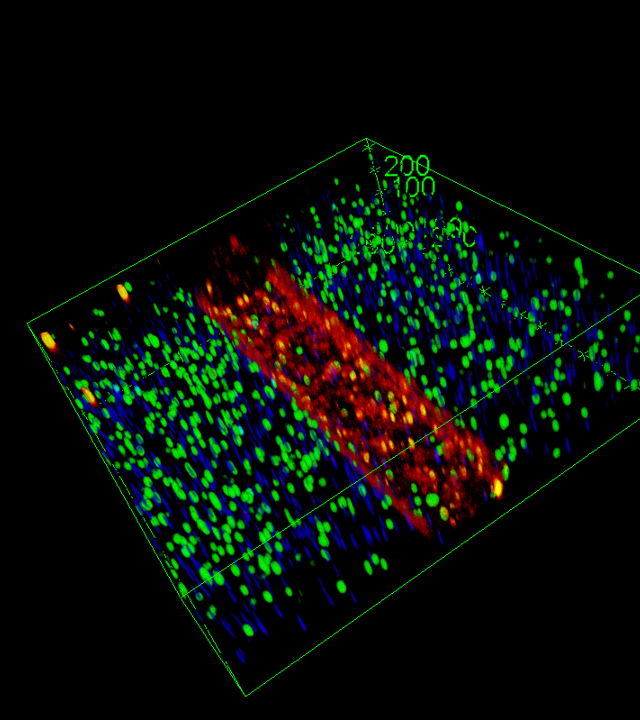
This platform will recapitulate the biology of the human optic nerve head—the exit structure of retinal ganglion cell axons and a region that is particularly vulnerable in primary open-angle glaucoma (POAG)—by unidirectionally connecting retinal organoids to cerebral assembloids through a three-dimensional channel that enables RGC axon growth and fasciculation. By allowing the reconstruction of a three-dimensional optic nerve, this device represents a major advance over existing two-dimensional models.
To faithfully reproduce the pathogenic environment of glaucoma, ON-Chip will incorporate an inflammatory component through the inclusion of glial cells and microglia—resident cells of the nervous system responsible for maintaining retinal homeostasis—as well as inflammatory monocytes, which are specifically observed in retinal degenerative conditions. ON-Chip will also allow controlled deformation of the three-dimensional channel to induce mechanical stress on RGC neuronal projections, thereby mimicking the mechanical stress caused by the abnormal elevation of intraocular pressure characteristic of POAG. Finally, retinal organoids may be genetically modified to introduce genetic variants identified in certain glaucoma patients.
By using genetically encoded sensors that enable real-time monitoring of retinal ganglion cell homeostasis, the platform will allow dynamic assessment of optic nerve functionality. By combining clinically relevant pathological conditions such as neuroinflammation or elevated intraocular pressure with patient-specific genetic risk factors, ON-Chip will constitute a unique and personalised model of glaucoma pathophysiology. This system will be particularly well suited for pharmacological screening, gene therapy validation and applications in regenerative medicine.

Expected outcomes
In summary, the ON-Chip platform represents a transformative step in glaucoma research, combining technological innovation with biological relevance to deepen our understanding of disease mechanisms and accelerate the development of targeted therapies.
The consortium
This ambitious project brings together three leading French neuroscience institutes: the Institut de la Vision, NeuroSpin (NeuroSU) and the Institut du Cerveau (ICM), which will contribute their complementary expertise in microfluidic engineering, organoid biology and neurodegenerative disease modelling.
Institut de la Vision; Institut du Cerveau; Sorbonne Université / IBPS (Neuro-SU)
Plus de projets