FERTILOoC
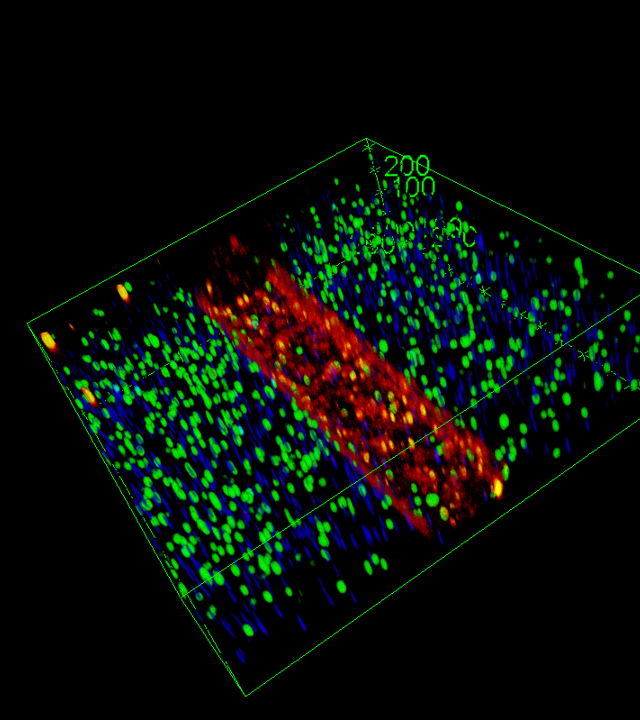
One in six couples worldwide experience infertility issues, and male infertility is responsible for about half of the cases, constituting a public health concern. Throughout male reproductive life, spermatogenesis originates from a small pool of spermatogonial stem cells (SSCs) that can self-renew or differentiate, undergo meiosis and finally spermiogenesis to produce sperm. In patients undergoing therapies with high gonadal toxicity, one of the major side effects that impairs the quality of life after recovery is the damage to the stock of SSCs and the resulting infertility issues. This point is especially critical for prepubertal boys who do not yet produce sperm. For fertility preservation, prepubertal boys are offered a cryopreservation of biopsy of the immature testicular tissue to preserve SSCs, in the hope to develop therapies allowing the derivation of sperm from these SSCs. In humans, the identification of the SSCs pool is not still defined, and the molecular mechanisms governing self-renewal and differentiation are barely known, hindering the understanding and improvement of the regenerative potential of SSCs which is a critical parameter for the successful development of new cell therapy from the SSCs present in the tissue.
Coordinator

Research Director
CEA
Institutions and establishments involved
Work Packages
CEA; Inserm
The scientific objectives are addressed through three work packages:
WP1 : Design and fabrication of an FCB-based organ-on-chip system for parallel analysis of several chips in different microfluidic conditions
WP2 : Prevalidation of OoC with testicular biopsies from mice
WP3 : Translation to human biopsy model
Programme de recherche
Testicular Organ-on-Chips (OoC) aim to recapitulate in vitro the microenvironment necessary to mature prepubertal testicular tissue, allowing SSCs to selfrenew and to induce spermatogenesis. These devices would provide a great model to help to delineate the human SSC pool, to unravel mechanisms governing their physiology, and their interactions with the niche in living human testicular tissue in vitro. This will undoubtedly contribute to their use in cell therapy in the context of fertility restoration. Testicular organ-on-chip would also benefit to the improvement of anticancer treatments protocols allowing to screen the effects of therapeutic molecules/physical agents on the pool of SSCs and testicular tissue functionality.


Expected outcomes
We aim to develop a new system of OoC allowing parallel live imaging analysis of several chips in different conditions of culture with high complexity microfluidics conditions, such as for example oscillation patterns. The elaboration of oscillatory fluidic patterns in nutriment supply is very important for the reproduction of the “systemic” environment of testicular tissue, such as the hormonal regulation. Parallelization is a critical point for organ-on-chip technology used in stem cells fate studies, because the scaling-up of the number of analyzable chips per experiment is necessary to obtain robust data. The design of such modular system based on parallelization and on live microscopic imaging is a promising tool to study stem cell physiology and interactions with the testicular niche. This approach applied to
murine and human testicular tissue should provide useful insights for both future basic research and medical applications of SSCs for treatment of infertility, contributing to a better clinical management of fertility preservation for cancer patient.
The consortium
We propose in this project to gather multidisciplinary teams, with expertise in basic and clinics research in reproduction and in design of chips and microfluidics, to engineer microfluidic devices to study the self-renewal and differentiation of SSCs in testicular biospies.
DRCM/Stabilité Génétique, Cellules Souches et Radiations; Institut Cochin; CEA-LETI
Plus de projets